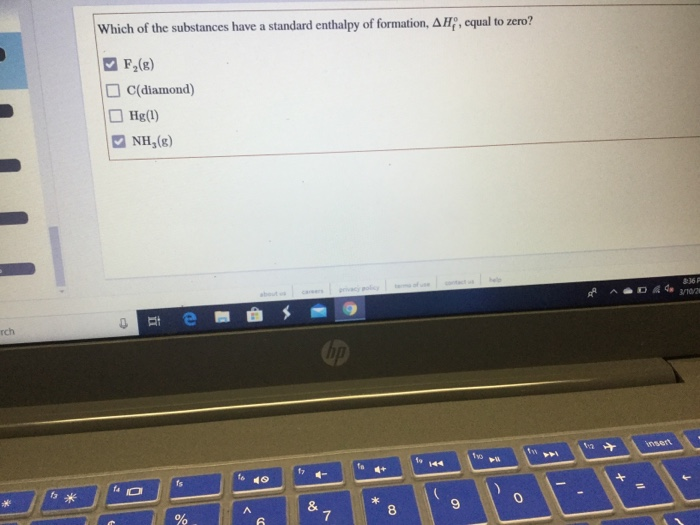
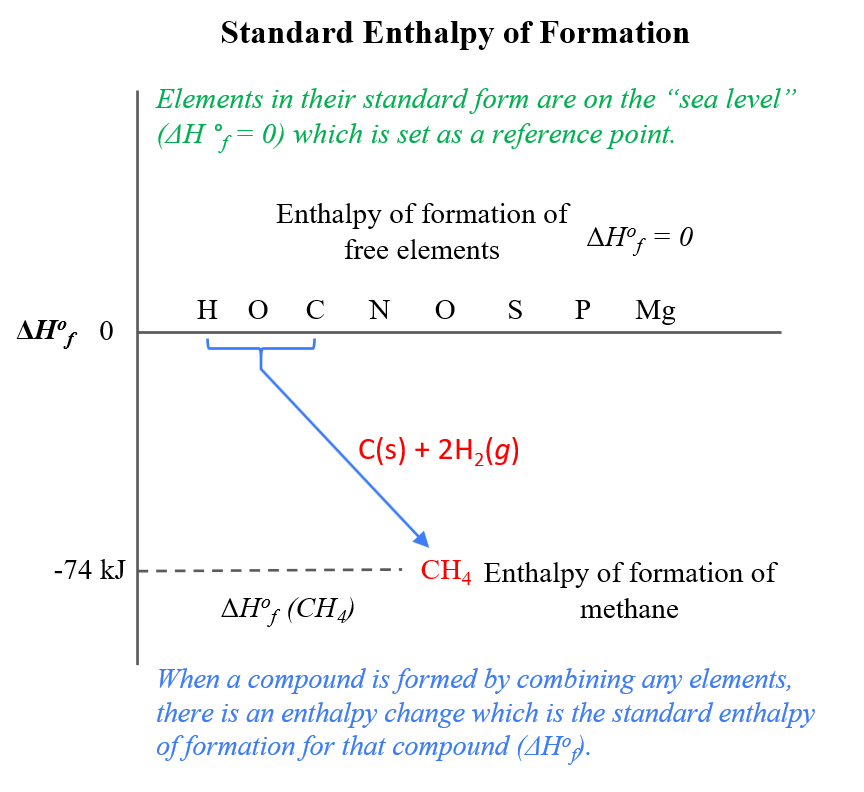
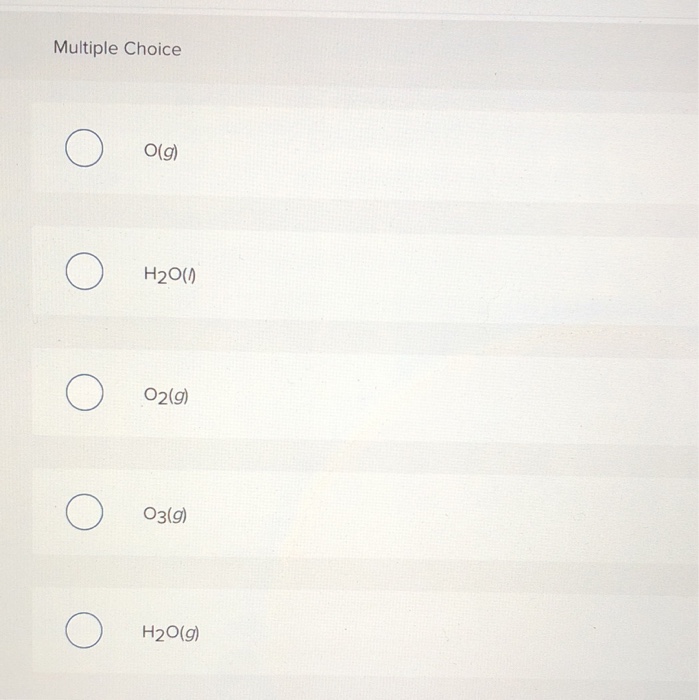
Which Substance Has A Standard Enthalpy Of Formation Of Zero - The enthalpy difference between graphite and. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. 1 the standard enthalpy of formation of an element in its standard state is zero. Carbon naturally exists as graphite and diamond. So standard enthalpy is zero for graphite. Pure elements in their standard state have a standard enthalpy of formation equal to zero. There is also p 4 and s 8 (which are not diatomic, but you get the point).
193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. Carbon naturally exists as graphite and diamond. Pure elements in their standard state have a standard enthalpy of formation equal to zero. So standard enthalpy is zero for graphite. There is also p 4 and s 8 (which are not diatomic, but you get the point). 1 the standard enthalpy of formation of an element in its standard state is zero. The enthalpy difference between graphite and.
So standard enthalpy is zero for graphite. Pure elements in their standard state have a standard enthalpy of formation equal to zero. 1 the standard enthalpy of formation of an element in its standard state is zero. There is also p 4 and s 8 (which are not diatomic, but you get the point). Carbon naturally exists as graphite and diamond. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. The enthalpy difference between graphite and.
Standard Enthalpy Of Formation Definition, Table, Equation, 46 OFF
193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. There is also p 4 and s 8 (which are not diatomic, but you get the point). 1 the standard enthalpy of formation of an element in its standard state is zero. The enthalpy difference between graphite and. Carbon naturally exists as graphite and.
Standard Enthalpy Formation
So standard enthalpy is zero for graphite. Pure elements in their standard state have a standard enthalpy of formation equal to zero. There is also p 4 and s 8 (which are not diatomic, but you get the point). Carbon naturally exists as graphite and diamond. The enthalpy difference between graphite and.
Standard enthalpy of formation and standard free energy of formation of
1 the standard enthalpy of formation of an element in its standard state is zero. So standard enthalpy is zero for graphite. There is also p 4 and s 8 (which are not diatomic, but you get the point). The enthalpy difference between graphite and. Carbon naturally exists as graphite and diamond.
Standard enthalpy of formation
Carbon naturally exists as graphite and diamond. The enthalpy difference between graphite and. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. There is also p 4 and s 8 (which are not diatomic, but you get the point). Pure elements in their standard state have a standard enthalpy of formation equal to.
Standard enthalpy of formation is zero for.
The enthalpy difference between graphite and. 1 the standard enthalpy of formation of an element in its standard state is zero. So standard enthalpy is zero for graphite. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. Pure elements in their standard state have a standard enthalpy of formation equal to zero.
Solved Which of the substances have a standard enthalpy of
1 the standard enthalpy of formation of an element in its standard state is zero. Carbon naturally exists as graphite and diamond. So standard enthalpy is zero for graphite. There is also p 4 and s 8 (which are not diatomic, but you get the point). 193 rows all elements in their reference states (oxygen gas, solid carbon in the.
Standard Enthalpy Of Formation Is Zero For Sulphur at Joseph Morehouse blog
Carbon naturally exists as graphite and diamond. There is also p 4 and s 8 (which are not diatomic, but you get the point). 1 the standard enthalpy of formation of an element in its standard state is zero. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. Pure elements in their standard.
Solved Select which of the following standard enthalpy of
So standard enthalpy is zero for graphite. 1 the standard enthalpy of formation of an element in its standard state is zero. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. Carbon naturally exists as graphite and diamond. There is also p 4 and s 8 (which are not diatomic, but you get.
Solved Which of the following has a standard enthalpy of
So standard enthalpy is zero for graphite. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. Pure elements in their standard state have a standard enthalpy of formation equal to zero. 1 the standard enthalpy of formation of an element in its standard state is zero. There is also p 4 and s.
SOLVED Which of the following has zero standard enthalpy of formation
1 the standard enthalpy of formation of an element in its standard state is zero. The enthalpy difference between graphite and. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. Pure elements in their standard state have a standard enthalpy of formation equal to zero. So standard enthalpy is zero for graphite.
Carbon Naturally Exists As Graphite And Diamond.
The enthalpy difference between graphite and. So standard enthalpy is zero for graphite. 193 rows all elements in their reference states (oxygen gas, solid carbon in the form of. 1 the standard enthalpy of formation of an element in its standard state is zero.
Pure Elements In Their Standard State Have A Standard Enthalpy Of Formation Equal To Zero.
There is also p 4 and s 8 (which are not diatomic, but you get the point).